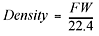Since gases are mostly empty space, the densities of gases are reported in g/L, not g/mL as found for solids and liquids. As you’re probably aware, density is equal to mass per unit of volume. To calculate the density of a gas at standard temperature and pressure, you take the molecular formula weight of the gas (grams per mole—from the periodic table) and divide that by the standard molar volume for a gas, which is 22.4 L per mole: