SAT2是SAT subject test的通常说法(学能倾向测验),SAT2考试有别于SAT1,它是一种专项考试,考察学生某一科目的能力,故又被称为SAT SUBJECT TESTS。
Sometimes atoms give their electrons up altogether instead of sharing them in a chemical bond. This process is known as disassociation. Water, for instance, dissociates by the following formula:
The hydrogen atom gives up a negatively charged electron, gaining a positive charge, and the OH compound gains a negatively charged electron, taking on a negative charge. The H+ is known as a hydrogen ion and OH– ion is known as a hydroxide ion.
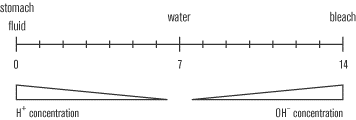
The disassociation of water produces equal amounts of hydrogen and hydroxide ions. However, the disassociation of some compounds produces solutions with high proportions of either hydrogen or hydroxide ions. Solutions high in hydrogen ions are known as acids, while solutions high in hydroxide ions are known as bases. Both types of solution are extremely reactive—likely to form bonds—because they contain so many charged particles.
The technical definition of an acid is that it is a hydrogen ion donor, or a proton donor, as hydrogen ions are consist of only a single proton. Acids put H+ ions into solution. The definition of a base is a little more complicated: they are H+ ion or proton acceptors, which means that they remove H+ ions from solution. Some bases can directly produce OH– ions that will take H+ out of solution. NaOH is an example of this type of base: